The Unanswered Question

Troponin Test
Thursday, June 21, 2012
The student question was, “My grandmother had a heart attack and received a troponin test, but I don’t understand why they test for a skeletal muscle protein in the blood?”
First off, troponin is more than just a single protein. It is in fact a 3-unit complex of troponin proteins known as I, T and C, (T stands for ‘tropomyosin binding’, I for ‘inhibitory’, and C for ‘calcium binding’). These three subunits are located at regular intervals along the length of actin filaments and play a key role in both skeletal and cardiac muscle contraction (but not smooth muscle). Apart from their proximity to each other in the troponin complex, troponin T, C and I are otherwise unrelated proteins with the different functions listed above.
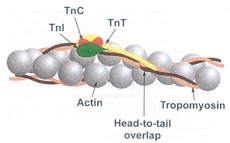
Figure: the three troponin subunits interacting with tropomyosin and actin in the thin filament.
Both cardiac and skeletal muscles are controlled by changes in intracellular calcium concentrations. When calcium rises, muscles contract, and when calcium falls, muscles relax. Troponin is a component of thin filaments (along with actin and tropomyosin), and is the protein to which calcium binds to accomplish this regulation (see figure). When calcium is bound to specific sites on TnC, tropomyosin rolls out of the way of the actin filament active sites, so that myosin (a molecular motor organized in muscle thick filaments) can attach to the thin filament and produce force and/or movement. In the absence of calcium, tropomyosin interferes with this action of myosin, and therefore muscles remain relaxed.
As I said above, troponin is found in both skeletal muscle and cardiac muscle, which is why the ‘why use a troponin test’ arose. As is turns out there are specific versions of troponin in the different types of muscle. The main difference is that the TnC subunit of troponin in skeletal muscle has four calcium ion binding sites, whereas in cardiac muscle there are only three. Diagnostically we can tell the difference between the types of troponin in a sample. In fact, cardiac troponin I and T are specific indicators of damage to the heart muscle (myocardium) that are released from damaged myocardium. They can thus be measured in the blood to differentiate between unstable angina and myocardial infarction (heart attack) in patients with chest pain or acute coronary syndrome. A patient who had suffered from a myocardial infarction would have an area of damaged heart muscle and consequently would have elevated cardiac troponin levels in the blood.
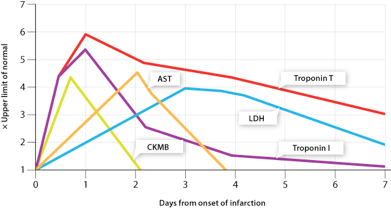
Troponins are now considered the biomarker of choice for detecting cardiac injury and have surpassed the historical ‘cardiac enzymes’ creatine kinase (CK), aspartate transaminase (AST) and lactate dehydrogenase (LDH), which are not specific to cardiac muscle (see top figure). Testing for cardiac troponin is associated with fewer false positive and false negative results.
Serum troponin concentrations begin to rise as soon as 3-4 hours following an acute myocardial infarction, and usually remain elevated for 7-10 days for TnI and 10-14 days for TnT. Results are typically not useful in the first one to three hours and maximum sensitivity is not until after 10 or more hours following onset of acute ischemia. It is also recognized that TnI is highly specific for cardiac muscle necrosis. The initial increase (3-4 hours) is due to troponin being released from the sarcoplasm, which is followed by the slower release of troponin from the cardiac myofilaments as they degrade. This pattern of release means that troponins can be detected in the serum for an extended period of time.
Comparison of troponin release with ‘traditional’ cardiac enzyme release patterns.


